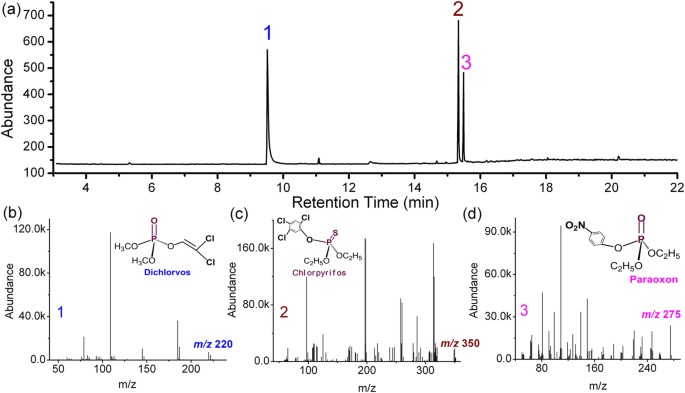
Molecules | Free Full-Text | Ethanol Determination in Post-Mortem Samples: Correlation between Blood and Vitreous Humor Concentration | HTML

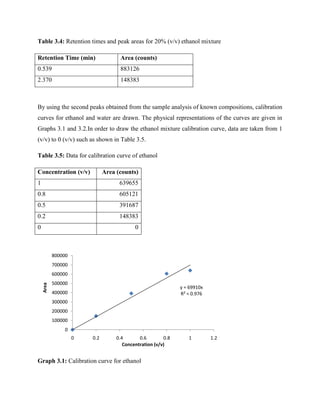
Table 1 from Improved Acetone-Butanol-Ethanol (ABE) Solution Analysis Using HPLC: Chromatograph Spectrum Deconvolution Using Asymmetric Gaussian Fit | Semantic Scholar

SOLVED:Question 5 3pE5 During analysis of a mixture of ethanol and propanol The chromatogram gave the following results for ethanol: retention time of 283 sec and half-height width of 3.6 seconds And

HPLC chromatogram of glibenclamide in methanol: ethanol (50:50 % v/v)... | Download Scientific Diagram
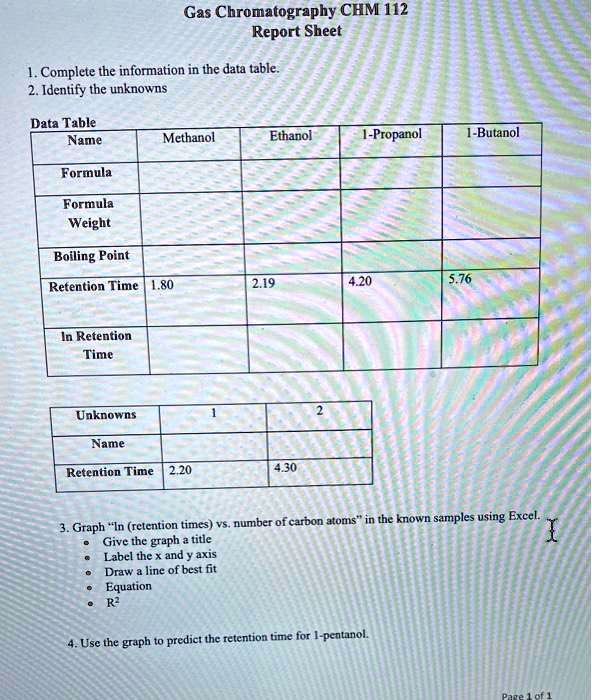
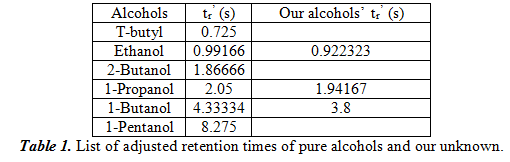
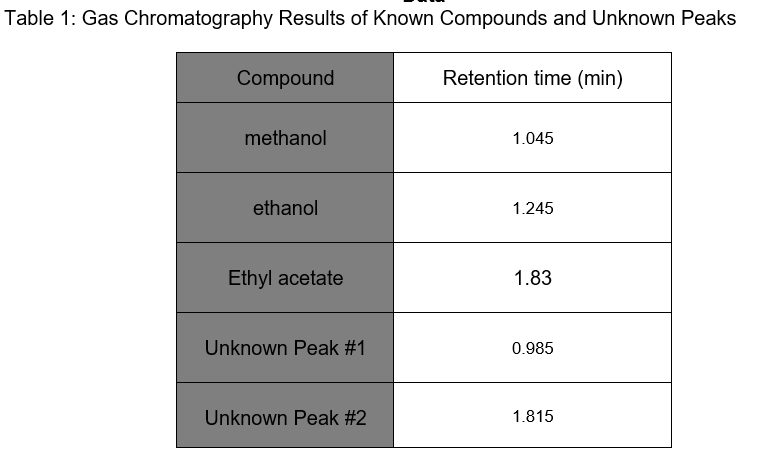
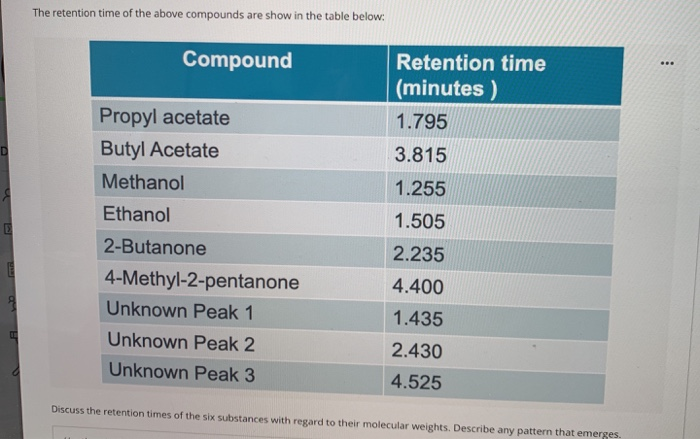
SOLVED:Gas Chromatography CHM 112 Report Sheet Complete the information in the data table 2. Identify the unknowns Data Table Name Methanol Ethanol [-Propanol Butanol Formula Formula Weight Boiling Point Retention Time 2.19
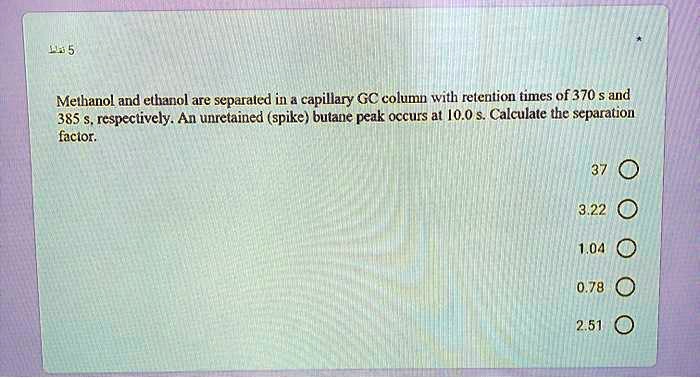
SOLVED:Methanol and ethanol are separated in & capillary GC column with retention times of 370 $ and 385 respectively: An unretained (spike) bulane peak occurs at /0.0 $ Calculate the scparation facior: 3.22 1,04 0.78 2.51

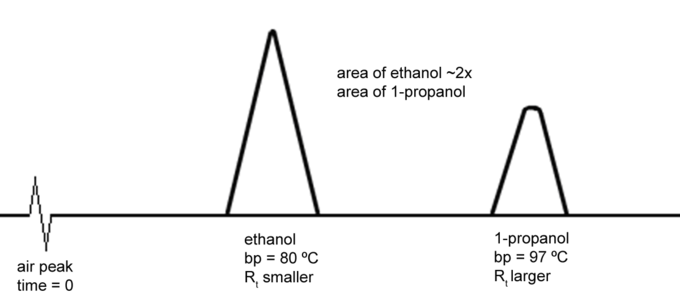
SOLVED:Amixture of ethanol (CzHsO) and butanol (CaHioO) is approximately 90% ethanol and 1O% butanol The mixture is passed through a gas liquid chromatogram: The printout obtained is likely to show that; compared
Topical anti-inflammatory activity of Polygonum cuspidatum extract in the TPA model of mouse ear inflammation | Journal of Inflammation | Full Text

GC-FID chromatogram for methanol (MeOH) and ethanol (EtOH), together... | Download Scientific Diagram

Solution Tutorial 3.pdf - Solution: KKEK2233 Physical and Analytical Chemistry II Lecturer: Dr. Awanis Tutorial 3 Date: 8th May 2018 1. Ethanol and | Course Hero
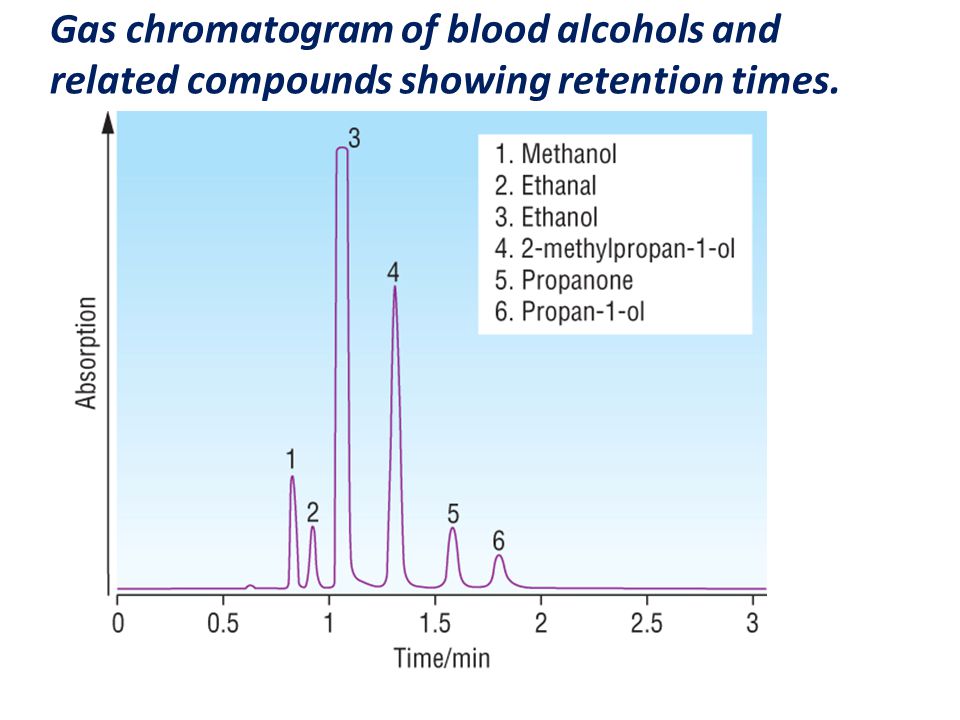
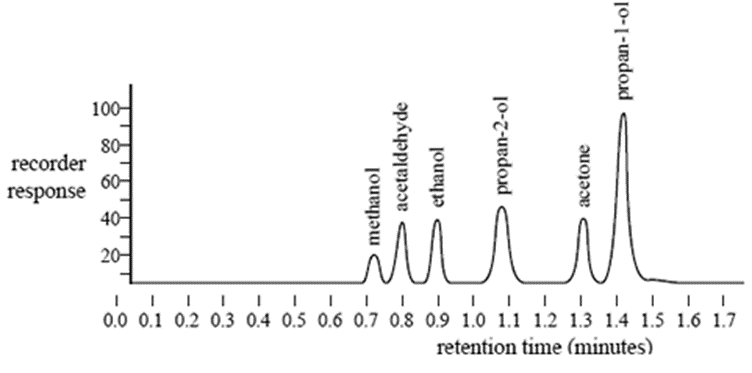
Gas Chromatography, GC L.O.: Explain the term: retention time. Interpret gas chromatograms in terms of retention times and the approximate proportions. - ppt download

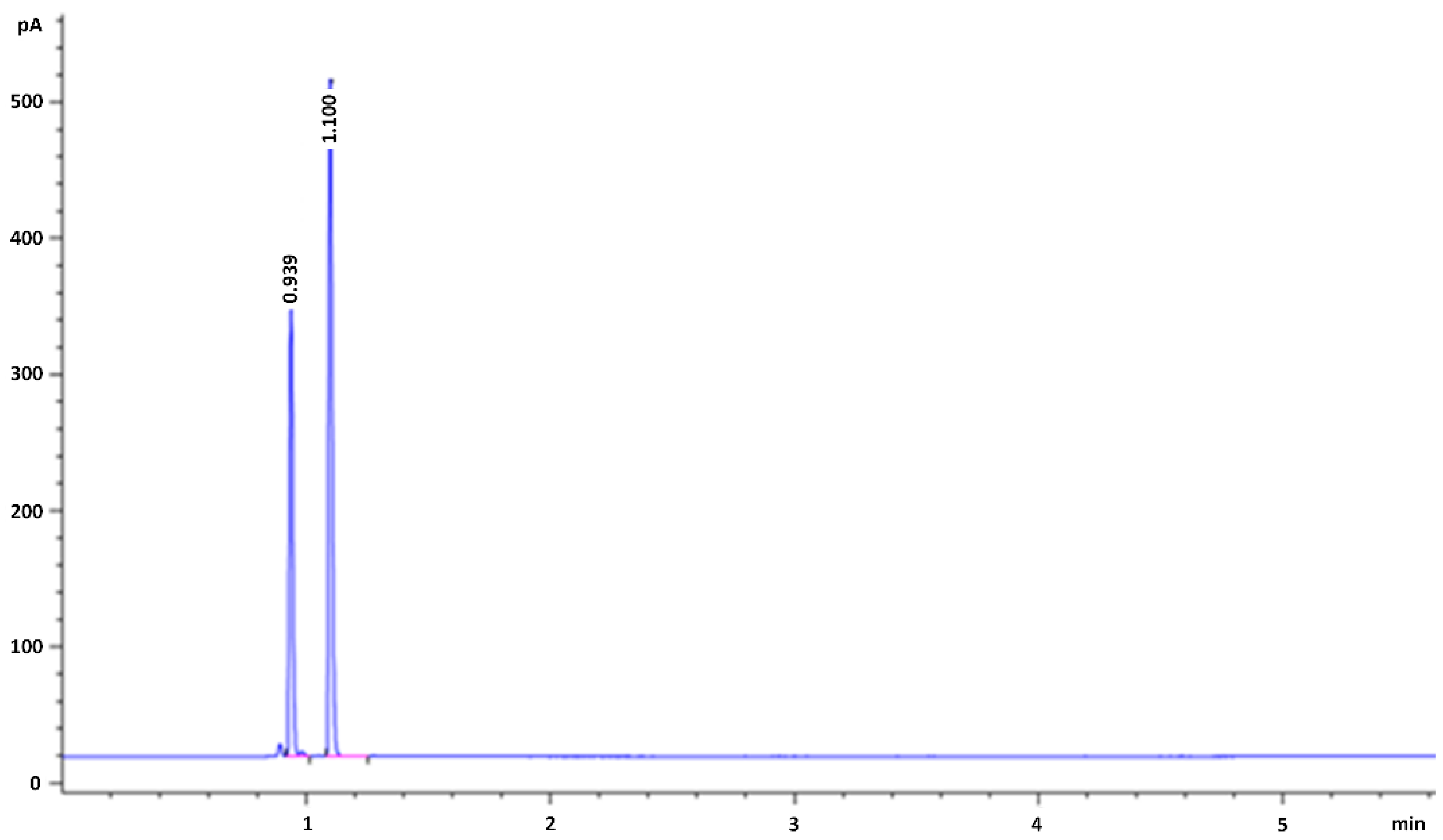
Rapid determination of ethanol in fermentation liquor by full evaporation headspace gas chromatography. | Semantic Scholar
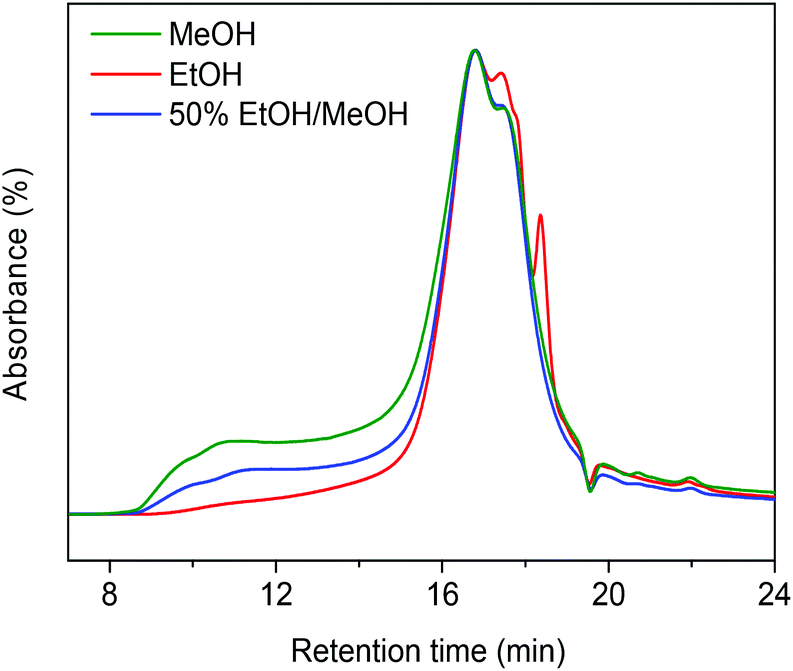
Ethanol as capping agent and formaldehyde scavenger for efficient depolymerization of lignin to aromatics - Green Chemistry (RSC Publishing) DOI:10.1039/C5GC01120E